 
This is the basis of an alternative (and more fundamental) definition of entropy: 
With more available microstates, the entropy of a system increases. In contrast to the macrostate, which characterizes plainly observable average quantities (temperature, for example), a microstate specifies all molecular details about the system, including the position and velocity of every molecule. For a given set of macroscopic variables, the entropy measures the degree to which the probability of the system is spread out over different possible microstates. The interpretation of entropy is the measure of uncertainty, which remains about a system after its observable macroscopic properties, such as temperature, pressure, and volume, have been taken into account. Thermodynamic entropy has the dimension of energy divided by temperature, which has a unit of joules per kelvin (J/K) in the International System of Units. As a result, entropy (denoted by S) is an expression of disorder or randomness. 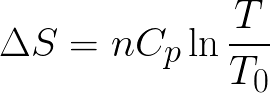
These processes reduce the state of order of the initial systems. It determines that thermal energy always flows spontaneously from regions of higher temperature to regions of lower temperature, in the form of heat. Therefore, entropy is also a measure of the tendency of a process, such as a chemical reaction, to be entropically favored or to proceed in a particular direction. In classical thermodynamics, the second law of thermodynamics states that the entropy of an isolated system always increases or remains constant. microstate: The specific detailed microscopic configuration of a system.Įntropy of the Playroom: Andrew Vanden Heuvel explores the concept of entropy while cleaning the playroom.entropy: A thermodynamic property that is the measure of a system’s thermal energy per unit temperature that is unavailable for doing useful work.Fundamentally, the number of microstates is a measure of the potential disorder of the system.The more such states available to the system with appreciable probability, the greater the entropy.The entropy of an isolated system always increases or remains constant. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
